 
He also had the great pleasure of seeing several of his close associates receive their own Nobel Prizes, including Rutherford in chemistry (1908) and Aston in chemistry (1922). Thomson received various honors, including the Nobel Prize in Physics in 1906 and a knighthood in 1908. In 1913 Thomson published an influential monograph urging chemists to use the mass spectrograph in their analyses. His nonmathematical atomic theory-unlike early quantum theory-could also be used to account for chemical bonding and molecular structure (see Gilbert Newton Lewis and Irving Langmuir). Ernest Rutherford, British physicist who discovered that the atom is mostly empty space surrounding a massive nucleus and who did many pioneering experiments with radioactivity. Of all the physicists associated with determining the structure of the atom, Thomson remained most closely aligned to the chemical community. The theory’s second component states that all atoms of a particular element have the same mass and characteristics. All matter is made up of indivisible atoms, according to the first component of his theory. He was a good lecturer, encouraged his students, and devoted considerable attention to the wider problems of science teaching at university and secondary levels. The atomic theory of Dalton was the first comprehensive effort to define all matter in terms of atoms and their characteristics. Even though he was clumsy with his hands, he had a genius for designing apparatus and diagnosing its problems. In the years after Dalton described his atomic model, multiple experiments were performed that proved that charged particles exist. In 1884 he was named to the prestigious Cavendish Professorship of Experimental Physics at Cambridge, although he had personally done very little experimental work. He was then recommended to Trinity College, Cambridge, where he became a mathematical physicist. Instead young Thomson attended Owens College, Manchester, which had an excellent science faculty. His father intended him to be an engineer, which in those days required an apprenticeship, but his family could not raise the necessary fee. 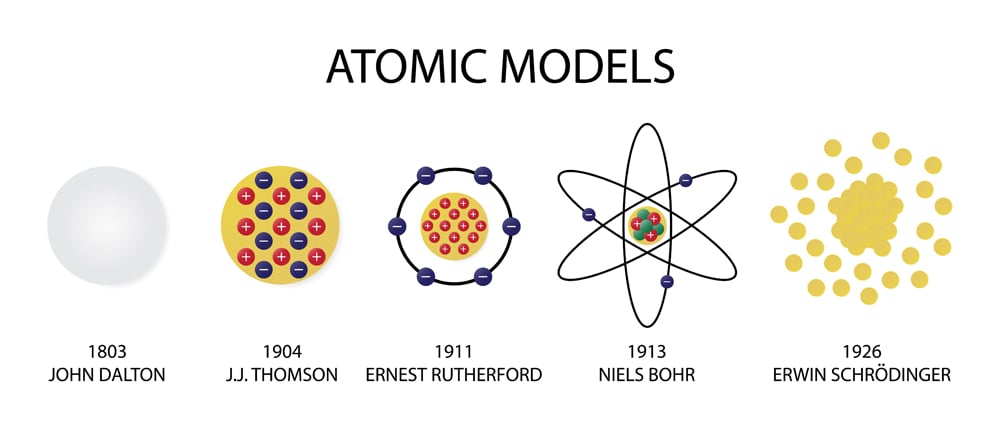
Ironically, Thomson-great scientist and physics mentor-became a physicist by default. His assistant, Francis Aston, developed Thomson’s instrument further and with the improved version was able to discover isotopes-atoms of the same element with different atomic weights-in a large number of nonradioactive elements. Here his techniques led to the development of the mass spectrograph. It provided a physical picture of how elements combine to form. His book A New System of Chemical Philosophy (Part I, 1808 Part II, 1810) was the first application of atomic theory to chemistry. Thomson’s last important experimental program focused on determining the nature of positively charged particles. Atom - Dalton, Bohr, Rutherford: English chemist and physicist John Dalton extended Proust’s work and converted the atomic philosophy of the Greeks into a scientific theory between 18. His efforts to estimate the number of electrons in an atom from measurements of the scattering of light, X, beta, and gamma rays initiated the research trajectory along which his student Ernest Rutherford moved. In 1904 Thomson suggested a model of the atom as a sphere of positive matter in which electrons are positioned by electrostatic forces. He resigned at age 34 and became a private tutor.Structure of the Atom and Mass Spectrography Dalton became a teacher of mathematics and natural philosophy (the study of nature and physics) at age 27 at a dissenting academy in Manchester. He could not attend an English university because he was a Dissenter (opposed to being required to join the Church of England), so he learned about science informally from John Gough, a mathematician and experimental physicist. Within just a few years, despite their lack of higher education, John and his brother started up their own Quaker school. John Dalton started working when he was 10 years old and began teaching at a local school at age 12. He learned from his father, a weaver, and from Quaker John Fletcher, who taught at a private school. That is, there must be some point beyond which we cannot go in the division of matter.I have chosen the word “atom” to signify these ultimate particles."ĭalton was born into a Quaker family on September 6, 1766. Notable Quote: "Matter, though divisible in an extreme degree, is nevertheless not infinitely divisible.Awards and Honors: The Royal Medal (1826), the fellowship of the Royal Society of London and the Royal Society of Edinburgh, honorary degree from the University of Oxford, associate of the French Academy of Sciences,.Published Works: New System of Chemical Philosophy, Memoirs of the Literary and Philosophical Society of Manchester.  
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
